Fundamentals of ion exchange
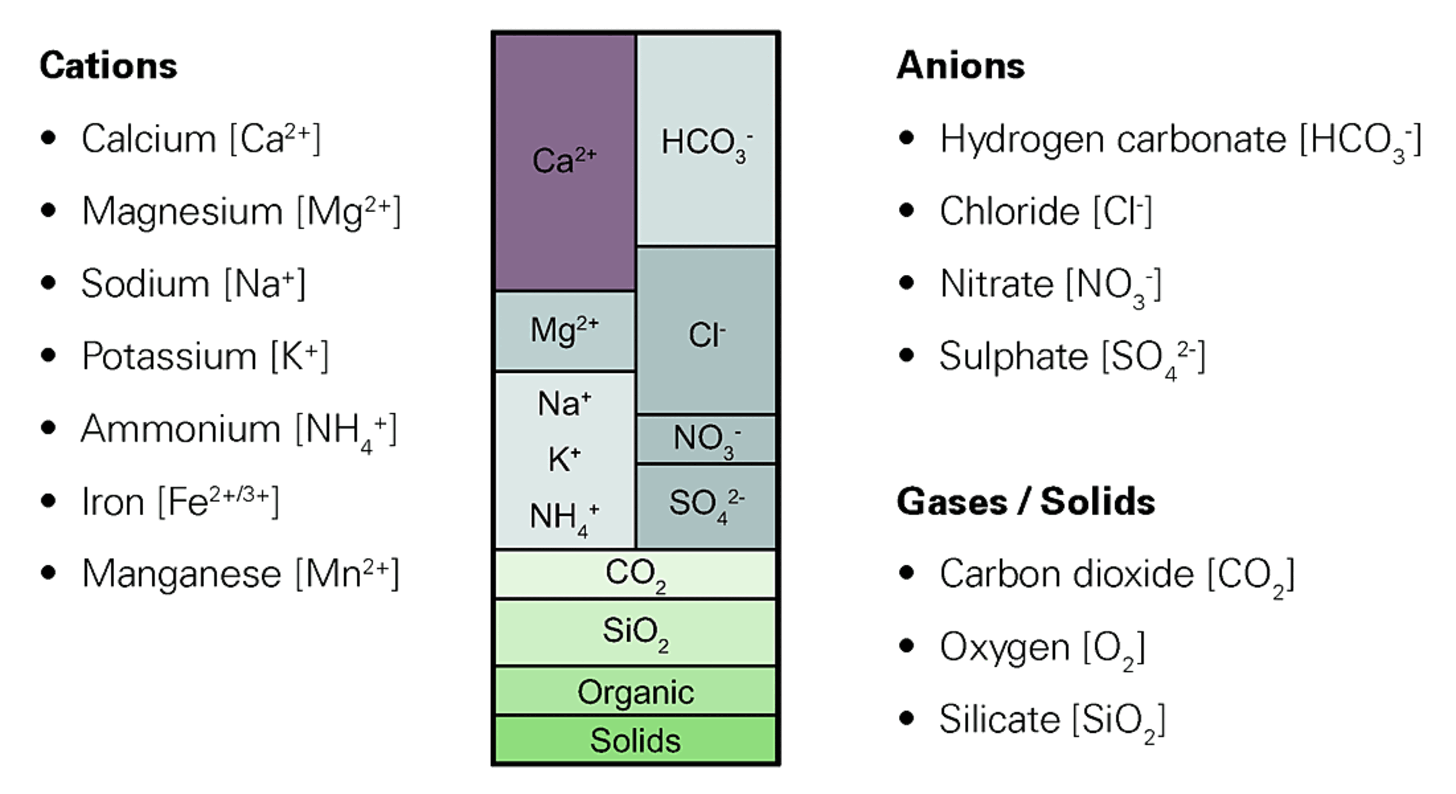
Water is a compound of hydrogen and oxygen that results from the combustion of hydrogen. It is an excellent solvent. For this reason, natural water does not exist as a chemically pure substance. It contains dissolved minerals, salts and organic compounds and also disperse and colloid disperse substances, like gases, in varying concentrations and compositions.
The dissolved salts are present as ions and can be removed from the water by means of special processes.
Ion exchange - principle
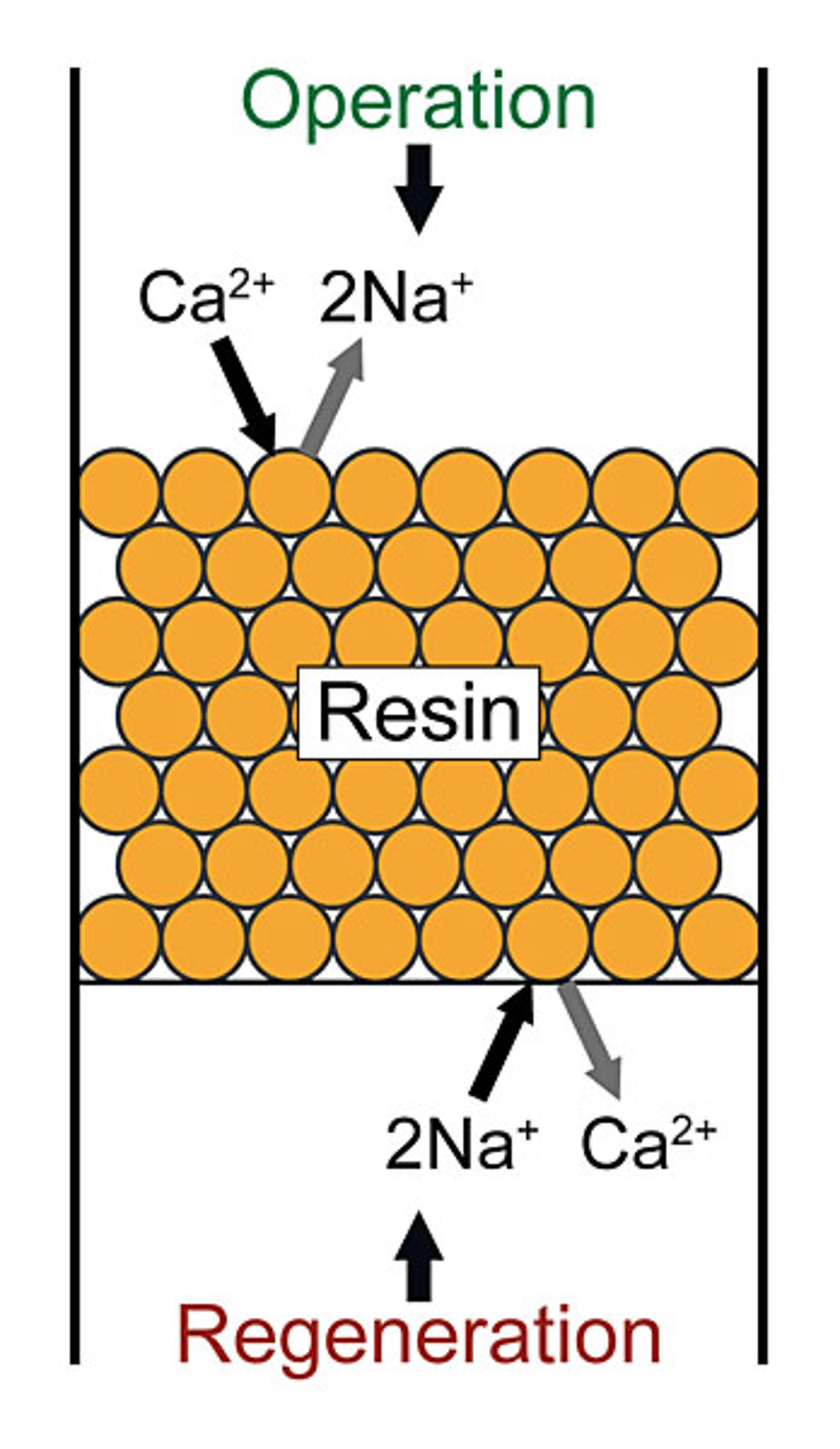
Ion exchangers are synthetic resins (organic polymers) into which various exchange-active groups, at which ions accumulate, are built. In the exchange process, the ions of the solution to be treated are exchanged with accumulated ions with the same electrical charge on the resin.
Basically, a differentiation is made between cation and anion exchangers. These types are further differentiated regarding the exchange-active groups:
- weak acidic / strong acidic
- weak basic / medium basic / strong basic
The terms "ion exchanger" and "resin" are used as synonyms when referring to water treatment.
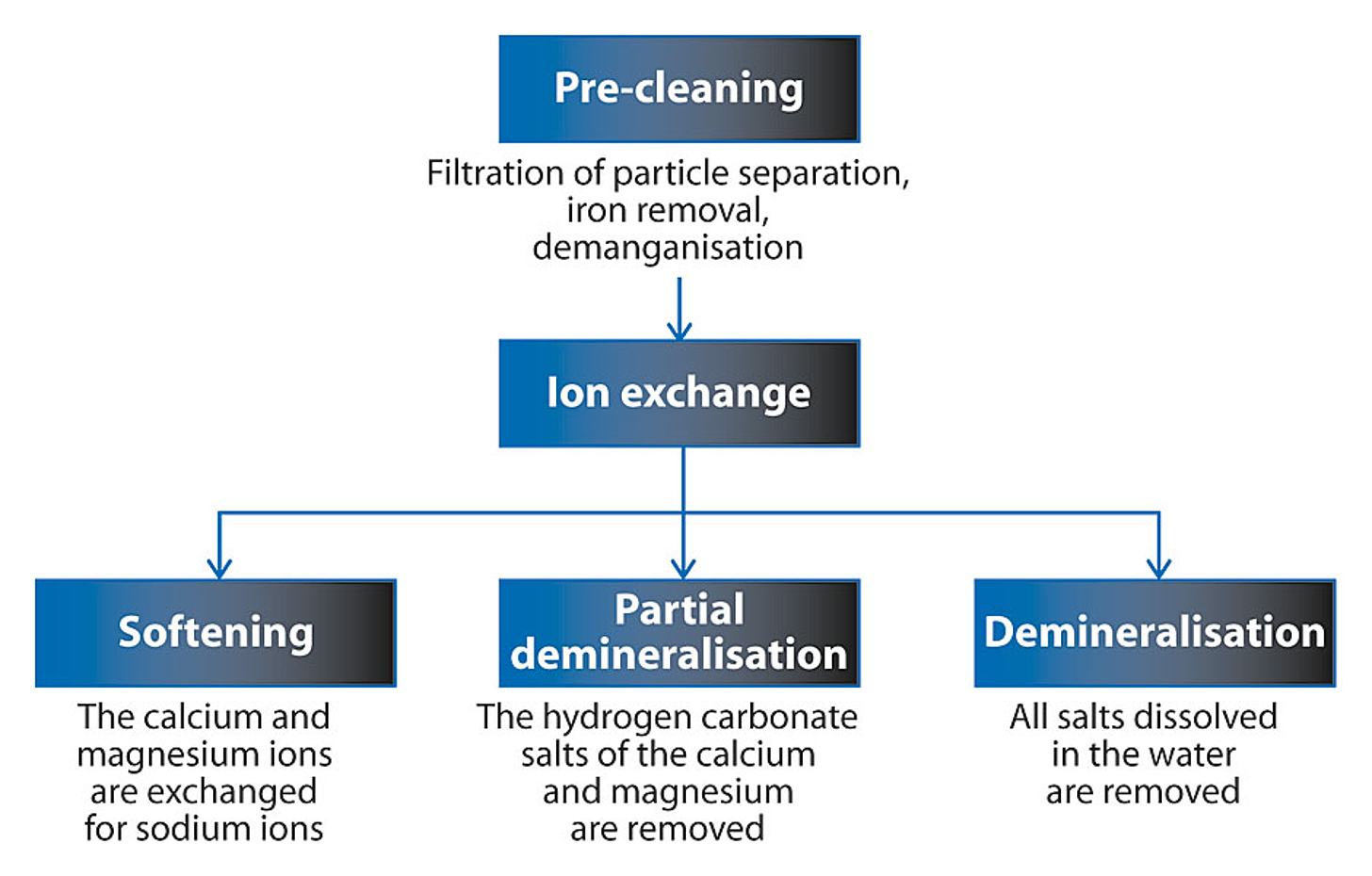
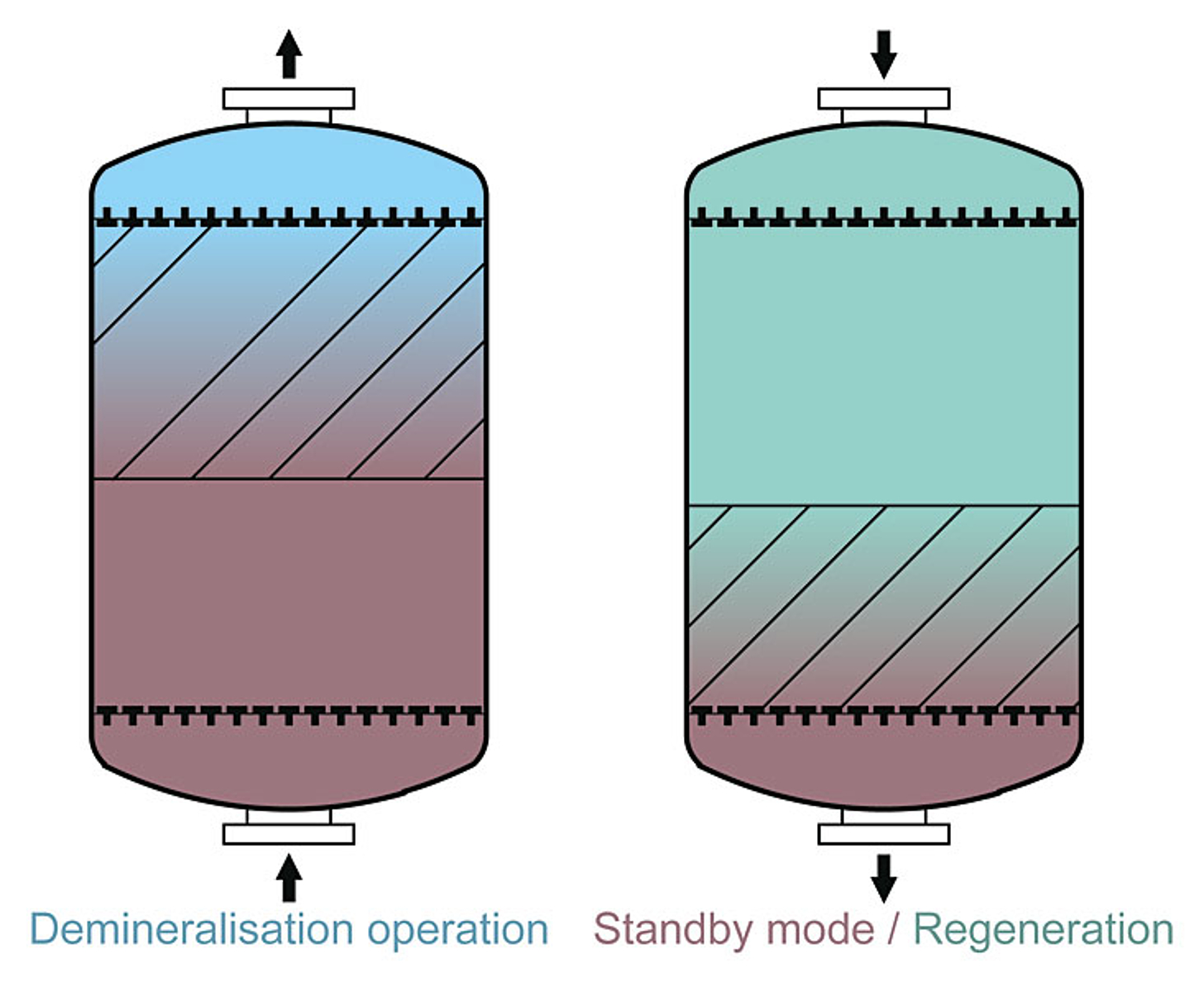
Floating bed process
Due to its many advantages, the floating bed method is mostly used for ion exchange systems.
Advantages:
- Extremely effective regenerant use combined with optimum demineralisation performance
- Maximum variability in the resin selection
- Low inherent water consumption
- Very compact tank design
Good pre-cleaning of the raw water is a prerequisite for using this process. The water must be free of iron and manganese, and must also be free of suspended substances.