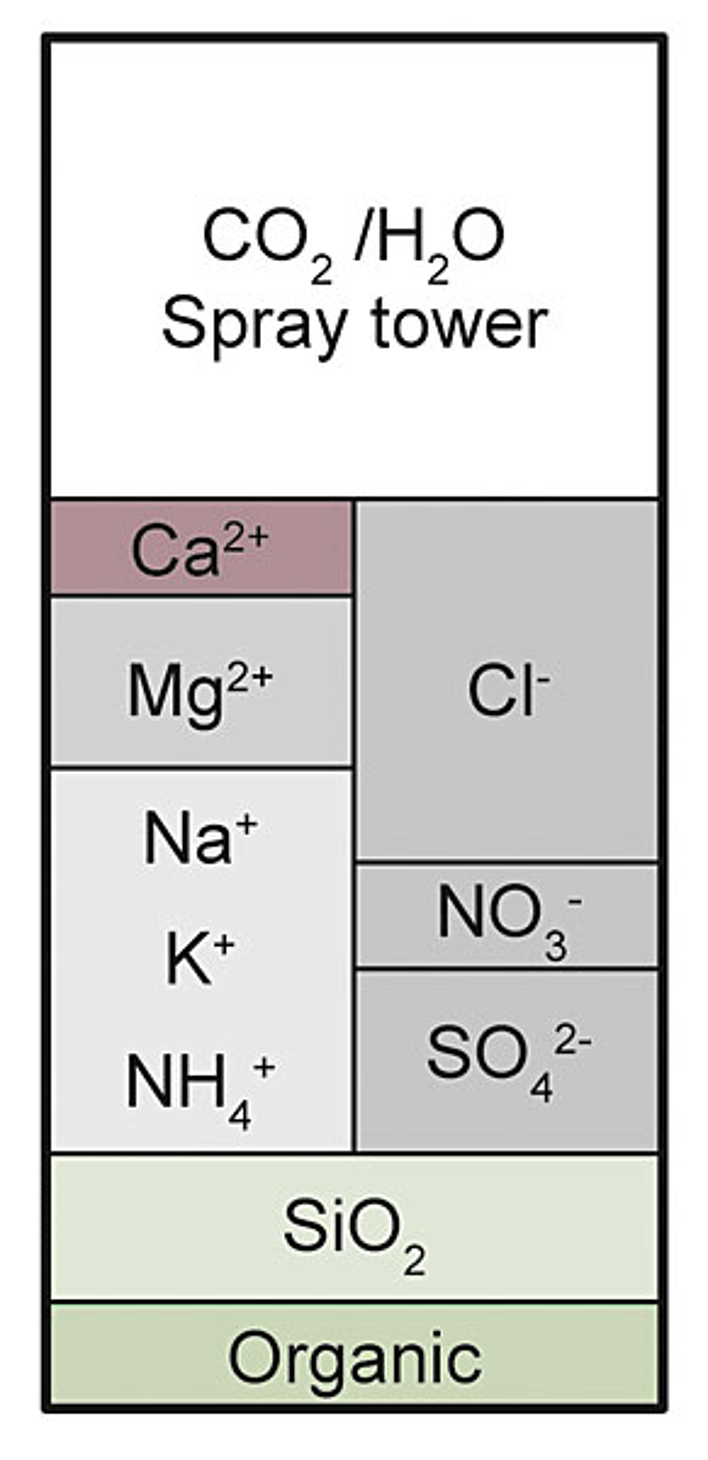
Decarbonisation/Partial desalination
With softening, only the ions of the hardeners calcium and magnesium are exchanged. With decarbonisation, on the other hand, the hydrogen carbonate salts of the calcium and magnesium, the so-called carbonate resins, are removed.
This process results in partial desalination of the water. Partial desalination occurs via filtration using a weakly acidic cation exchanger loaded with hydrogen ions.
Carbon dioxide results from decarbonisation and has to be removed in a downstream percolator. In many cases, decarbonisation is preceded by softening. Regeneration primarily occurs with hydrochloric acid.
The graphic shows the water matrix after decarbonisation.