Partial desalination
With softening, only the ions of the hardeners are exchanged. With decarbonisation, on the other hand, the hydrogen carbonate salts of the calcium and magnesium, the so-called carbonate resins, are removed.
This process results in partial desalination of the water. Partial desalination occurs via filtration using a weakly acidic cation exchanger loaded with hydrogen ions.
Carbon dioxide results from decarbonisation and has to be removed in a downstream percolator. In many cases, decarbonisation is preceded by softening. Regeneration primarily occurs with hydrochloric acid.
Demineralisation
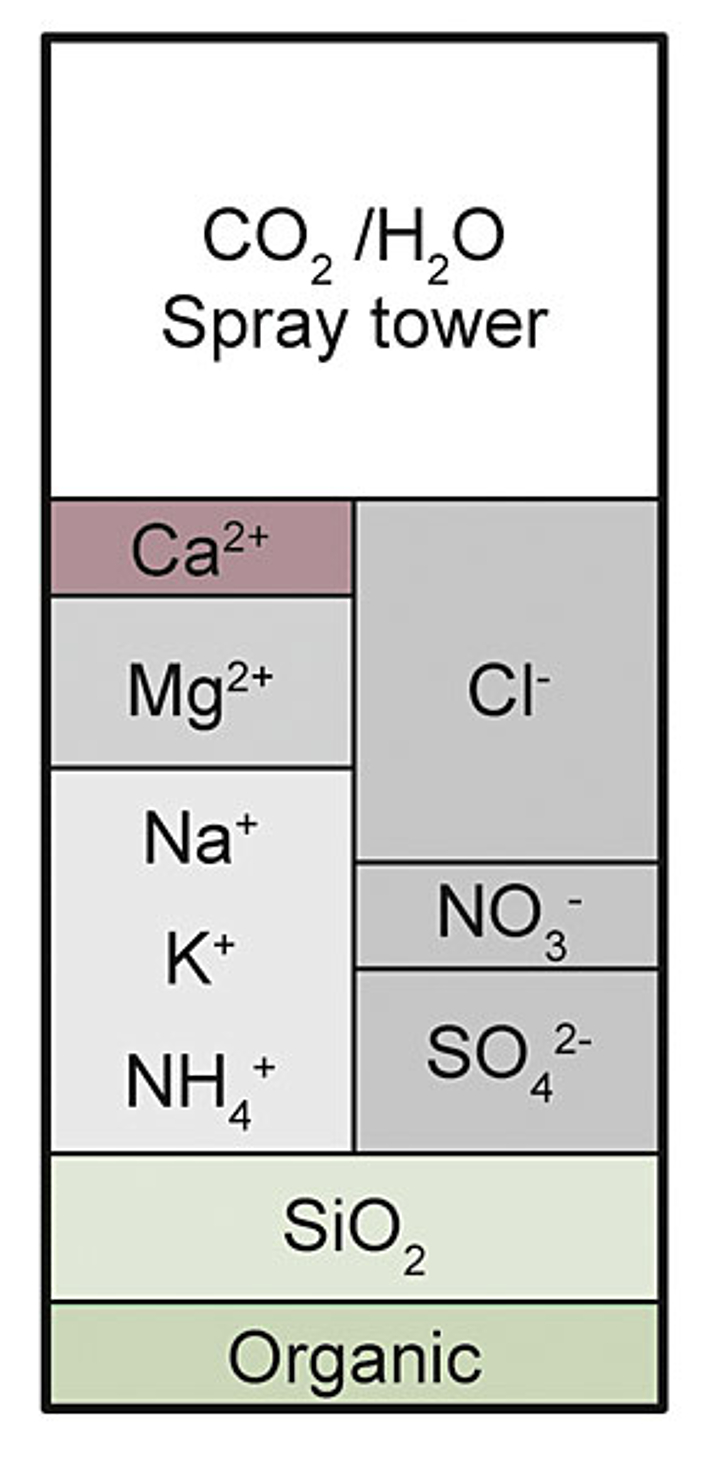
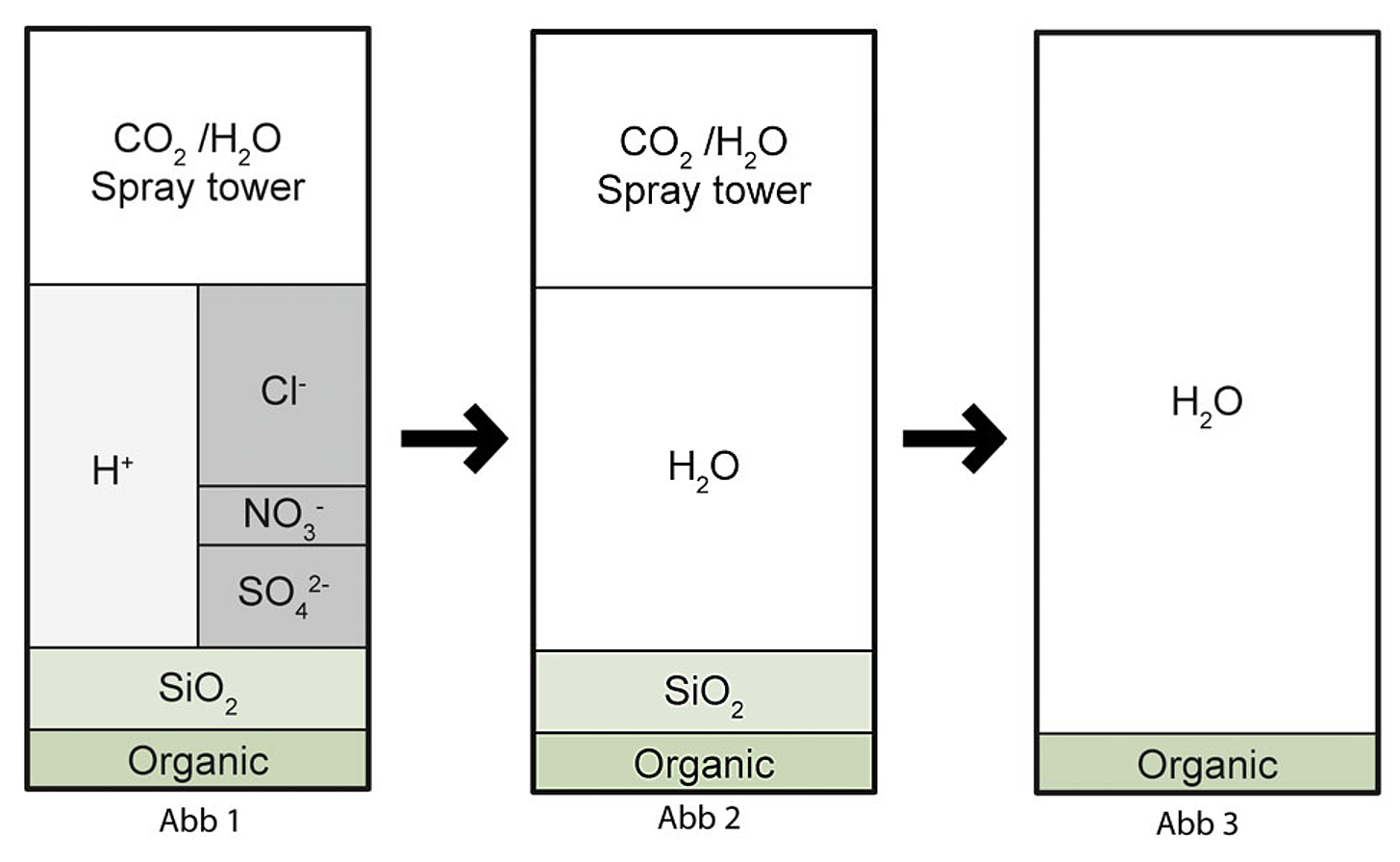
With demineralisation, all salts dissolved in the water are removed through a combination of strongly acidic cation exchangers and strongly basic anion exchangers. In the first desalination stage, all cations are exchanged for hydrogen ions. The concentrations of anions remain unchanged, i.e. strong acids such as hydrochloric acid HCl, nitric acid HNO3 and sulphuric acid H2SO4, in addition to CO2, arise. This process is also called decationisation for this reason (see Fig. 1 (Abb 1)).
Regeneration of cation exchangers primarily occurs with hydrochloric acid.
The acidic discharge from the cation exchanger is passed over an anion exchanger in the second stage. If a weakly basic exchanger is used, only the mineral acids are removed (HCl, H2SO4, HNO3) (see Fig. 2 (Abb 2)).
If, on the other hand, a strongly basic exchanger is used, silicic acid can also be removed. The result is demineralised water (see Fig. 3 (Abb 3)). The regeneration of these exchangers occurs with strong bases, e.g. sodium hydroxide.